Introduction
Myostatin-Related Muscle Hypertrophy is characterized by an increase in muscle build up as well as muscle strength. People with Myostatin-Related Muscle Hypertrophy also have a lower fat body content. These symptoms are most noticeable in babies as young as 3 months old, up to young children because it is such a drastic difference between normal babies and children. Due to the extremely low frequency of this disease and diagnosis, it is unknown what percent of babies are born with Myostatin-Related Muscle Hypertrophy in the United States as well as globally. However, there are approximately seven known cases of people living with this disease in North America [1].
Currently, there is no treatment available for Myostatin-Related Muscle Hypertrophy. This is mostly due to its low frequency of occurrence in individuals as well as the belief that it is non-life-threatening. For these reasons, limited research has been done on this disease and its link to other diseases as well clinical implications of Myosatin -Related Muscle Hypertrophy [1].
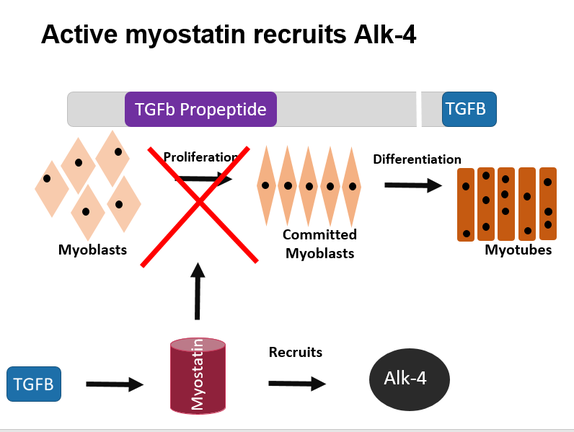
What is known about Myostatin-Related Muscle Hypertrophy is that it is controlled by the MSTN gene. MSTN has two domains: the TGFB propeptide and TGFB. TGFB is activated when it is cleaved from the rest of the gene. This then works as a transcription promotor to synthesize the protein myostatin. Myostatin works directly to stop the differentiation of myoblasts into committed myoblasts, which then differentiate into mature skeletal muscle cells. When the MSTN gene is dysfunctional, myostatin is not synthesized, thus allowing muscle cells to mature without regulation [2].
MSTN is highly conserved among many homologs, making this convenient to study with model organisms, especially the mouse. The mice phenotype and genotype are like those in humans and are easy to maintain in a lab setting, making them perfect to study Myostatin-Related Muscle Hypertrophy in humans. The gap in research that I would like to study in relation to this gene is its connection with lung cancer. About 1/3 of people with Myostatin-Related Muscle Hypertrophy develop lung cancer in their lifetime, making it an excellent target of further research. I believe this is due to the fact that, when present, myostatin also recruits Alk-4, which is in charge of regulation of cell differentiation, proliferation and apoptosis.
AIM 1
The first AIM is to link a sequence in MSTN to the recruitment of Alk-4. I hypothesize that the most conserved regions will be crucial to Alk-4 recruitment. To study this in my model organism, I will first use Clustal Omega to align the sequences of MSTN in different homologs and the human species. This would make sorting and the determination of conserved sequences easy. I would then use CRISPR/Cas9 to knockout those regions in mice. I will screen for tumors in the lungs in conjunction with a dysfunctional MSTN gene. I will use these mice for the rest of the experiment.
AIM 2
In my second AIMS, I will utalize RNA-seq as well as GO to determine allele-specific variations in transcription. I hypothesize that all genes controlling recruitment of Alk-4 in the lungs as well as other genes regulating cell proliferation and differentiation will be downregulated in the lungs due to the mutated MSTN gene. These downregulations are directly related to tumor development in the lungs.
AIM 3
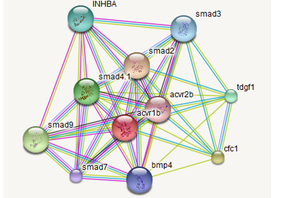
In the third AIM, I would determine the differences in protein-protein interactions when ALK-4 is present in the lungs and when it is absent in the lungs due to the lack of recruitment from myostatin. I hypothesize that with the loss of Alk-4, cells will not have a regulating protein to interact with, thus losing regulation interactions. I would expect the interactions to predictably decrease. This can help us understand how many proteins are effected by Alk-4.
Future Directions
In the future, I would like to perform a similar set of assays as the ones described above, only with the activated TGFB portion of the MSTN gene. This would help determine if lung tumors due to a dysfunctional MSTN gene is solely because of the absence of Alk-4 or if there are other factors that contribute to lungs cancer development.
References
[1] myostatin-related muscle hypertrophy - Genetics Home Reference
https://ghr.nlm.nih.gov/condition/myostatin-related-muscle-hypertrophy#synonyms
[2]Medical Definition of Myostatin
http://www.medicinenet.com/script/main/art.asp?articlekey=33546
https://ghr.nlm.nih.gov/condition/myostatin-related-muscle-hypertrophy#synonyms
[2]Medical Definition of Myostatin
http://www.medicinenet.com/script/main/art.asp?articlekey=33546


